

A recent research paper published in Journal of Agricultural and Food Chemistry describes an alternative pathway to the formation of trans-cinnamic acid (CA) derived from L-phenylalanine (L-Phe) in tea (Camellia sinensis) plants and other plants.
The researchers from South China Botanical Garden of Chinese Academy of Sciences found that CA is a precursor of many phenylpropanoid compounds, including catechins and aroma compounds, in tea (Camellia sinensis) leaves, and is derived from L-Phe deamination. Therefore, investigation on CA biosynthesis is of significance.
They have discovered an alternative CA formation pathway from L-Phe via phenylpyruvic acid (PPA) and phenyllactic acid (PAA) in tea leaves through stable isotope-labeled precursor tracing and enzyme reaction evidence. Both PPA reductase genes (CsPPARs) involved in the PPA-to-PAA pathway were isolated from tea leaves and functionally characterized in vitro and in vivo.
CsPPAR1 and CsPPAR2 transformed PPA into PAA and were both localized in the leaf cell cytoplasm. Rosa hybrida flowers (economic crop flower), Lycopersicon esculentum Mill fruits (economic crop fruit), and Arabidopsis thaliana leaves (leaf model plant) also contained this alternative CA formation pathway, suggesting that it occurred in most plants, regardless of different tissues and species.
These results uncover an alternative pathway to CA from L-phenylalanine in many plants, and improve our understanding of CA biosynthesis in tea plants and other plants.
For further reading, please refer to: https://pubs.acs.org/doi/10.1021/acs.jafc.9b07467.
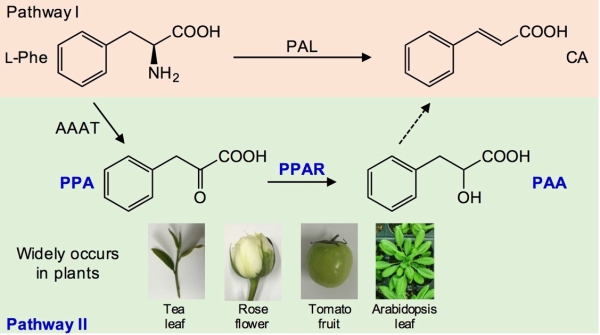
Figure. Alternative pathway for the formation of trans-cinnamic acid (CA) derived from L-phenylalanine (Phe) in plants.

